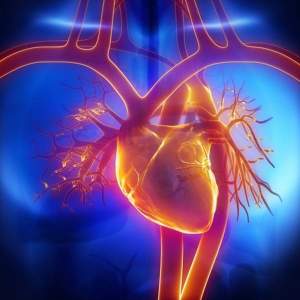
HeartWare Ventricular Assist System Approved
On November 20, the Food and Drug Administration announced the approval of the HeartWare Assist System that helps support heart function and blood flow for patients who are at the end-stage of heart failure and currently await a transplant.
The newly approved Assist System is a left ventricular assist device (LVAD)—the most common ventricular assist devices—but the newly approved LVAD is unlike other assist devices in the past. Former LVADs come with components that need to be planted in the patient’s abdomen, but the new HeartWare System is small enough to fit in the chest cavity near the heart. The new system provides specific advantages to smaller patients who cannot receive the abdomen implantation.
The HeartWare LVAD was approved after a clinical trial called the ADVANCE trial. 137 patients with advanced heart failure were analyzed and compared to other patients in the Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS). The advantages of the HeartWare LVAD showed more benefits than complications.
There are serious possible complications associated with the new LVAD though. Serious infections and strokes can occur when using the new HeartWave LVAD.
The approval of the new LVAD is the first time the FDA has approved an LVAD with the INTERMACS system as a control. The system is managed by the University of Alabama and represents a joint effort of the FDA, National Heart, Lung and Blood Institute (NHLBI), the Centers for Medicare and Medicaid Services (CMS), as well as scientists and surgeons.
Christy Foreman with the FDA’s Center for Devices and Radiological Health, stated: “Well-designed registries in targeted product areas can enhance the public health and provide a cost-effective approach to clinical research for industry innovators. For HeartWare, registry data directly facilitated the development and availability of this new device.”
Source: Food and Drug Administration
Comments
Must Read
 Pure Food and Drug Act
Pure Food and Drug ActPURE FOOD AND DRUG ACT TEXT What is the Pure Food and Drug Act? The Pure Food and Drug act of 1906 is a federal law that mandates for the inspection of meat products and forbids the sales, manufacturing or transportation of poisonous patent medicines and adulterated food products.
 4 Facts that you Must Know about Drug Possession
4 Facts that you Must Know about Drug PossessionDrug Possession DefinedA charge furnishing details expressing the illegal possession of Drugs may be defined as the unlawful, illicit, and prohibited ownership of Drugs; however, the nature within which the individual suspect in question gained access to the Drugs may also be associated with the investigation.
 Prempro
PremproWhat is Prempro? Prempro is a dangerous drug that contains medroxyprogesterone and a combination of conjugated estrogens (a mixture of estrogen hormones).
 Amphetamine
Amphetamine What is an Amphetamine?Amphetamines are psycho-stimulant drugs--within the phenethylamine class—that produce increased focus and wakefulness in association with decreased appetite and fatigue.
 Everything to know about Adderall
Everything to know about AdderallWhat is Adderall?Adderall is a form of medication used for attention-deficit hyperactivity disorder and narcolepsy.